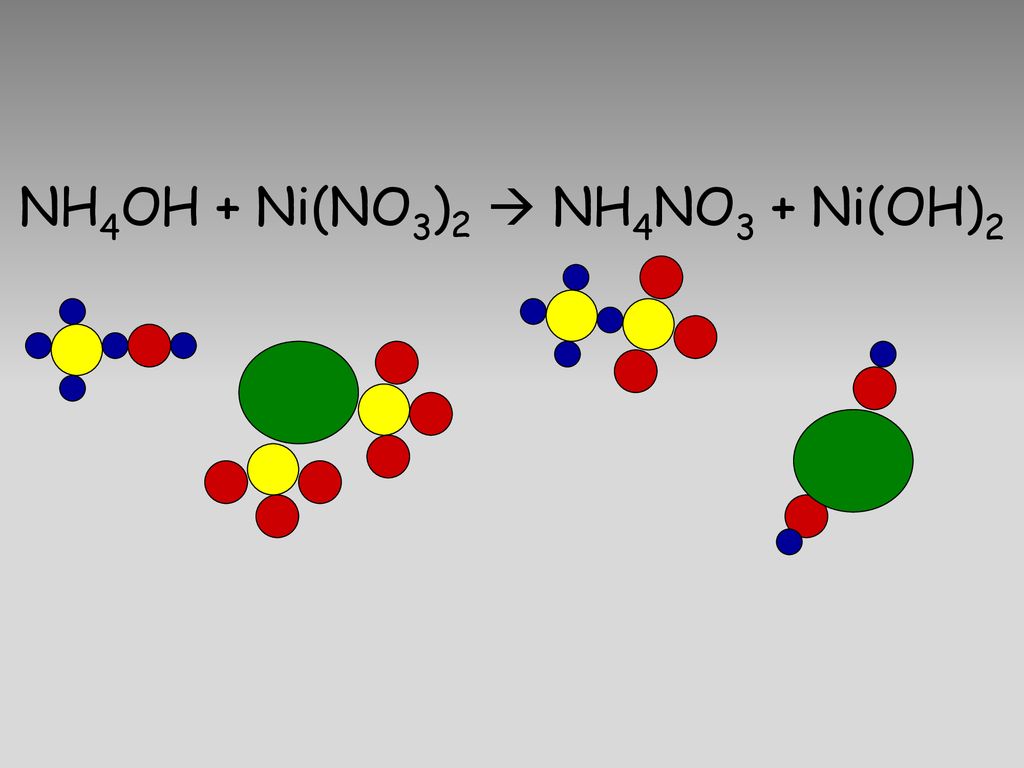
When dimethylglyoxime solution is added to an aqueous solution of nickel (II) chloride followed by ammonium hydroxide, then which of the following statements are incorrect?This question has multiple correct questions(a) No precipitate
29. NiCl2 + NH4OH + dimethylglyoxime ——> A ( complex ) Incorrect statement for complex A is /are 1 Coordination number of metal ion is 4 2 Two five membered and two
What is meant by aqueous ammonia solution? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium

Effect of temperature on Ni and Cd leaching recovery (NH4OH / (NH4)2CO3... | Download Scientific Diagram
What is nh3 aqueous solution?. NH3 aqueous means aqueous solution of… | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium

Effect of time on Ni and Cd leaching recovery (temperature=45 °C, NH4OH... | Download Scientific Diagram

Scanning Electron Microscope image of Ni-BTC MOFs. (a) Ni-BTC Anl ; (b)... | Download Scientific Diagram

SOLVED: When an aqueous solution of NH4OH is mixed with an aqueous solution of Ni(NO3)2, a pale yellow precipitate forms. Write a balanced molecular equation for this reaction. Write the complete ionic

SOLVED: A nickel coordination compound, Ni(NH3)6Cl2, can be prepared by a procedure similar to the one you will use to prepare Cu(NH3)4SO4·H2O. The reaction can be represented stoichiometrically as NiCl2·6H2O + 6
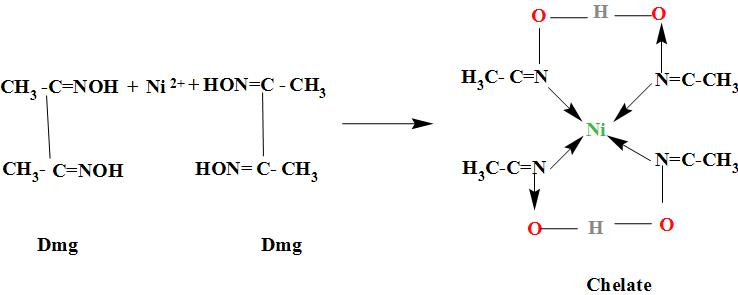
27. DMG +NiCl2+NH4OH makes Complex a+ NH4Cl+H2O. What is complex a and find the hybridisation magnetic character and Oxidation state of Nickel in complex a ?

NiCl_2 in the presence of dimethyl glycoxime (DMG) gives a complex which precipitates in the presence of NH_4OH, giving a bright red color.Draw its structure and show H bonding.

It is an experimental fact that:- DMG+Ni(II) salt+NH_4OH→ Red precipitate Which of the following ... - YouTube

Epitaxial N–H-doped Ni1−x O and Ni2O3 with special planar defects by pulsed laser ablation of metallic Ni in aqueous ammonia | Applied Physics A






/NH4OH%20Fusion%20Chemical%20Blending%20System.jpg?width=1305&name=NH4OH%20Fusion%20Chemical%20Blending%20System.jpg)